
Oregon Board Must Produce Plan by September That Could Lead to Price Caps
The battle to limit drug prices has reached the trenches in Oregon as the Oregon Prescription Drug Affordability Board moves forward on developing a plan due in September. The stakes are significant because the plan could be a blueprint for price caps considered by the 2025 legislature.
 According to The Lund Report, which focuses on Northwest health issues, pharmaceutical industry lobbying against the prospect of drug price caps has intensified. “Pharma lobbyists have an endless and infinite bench of resources and that can be difficult to go up against,” says Ralph Magrish, the board’s executive director.
According to The Lund Report, which focuses on Northwest health issues, pharmaceutical industry lobbying against the prospect of drug price caps has intensified. “Pharma lobbyists have an endless and infinite bench of resources and that can be difficult to go up against,” says Ralph Magrish, the board’s executive director.
Drug makers claim the board is ill-suited to its assigned task. They persuaded U.S. District Court Judge Michael Mosman to block the board from requiring drug makers to justify price increases. And, they have aligned with similar board criticism by patient groups that receive drug industry support.

Reid Porter, senior public affairs director for PhRMA, questions whether the state board is up to the job it’s been given. “Frankly, there seems to be political points that were scored by establishing the board,” he told The Lund Report.
The impetus for creating the board has been a steep increase in the price of prescription drugs amid reports the same drugs sell for far less in other countries with price controls. A report released in 2022 showed prescription drugs rose more between 2013 and 2019 than any other health care cost. In that time period, commercial insurers’ per-person spending on pharmaceutical drugs nearly doubled to more than $1,000 per year.
“We firmly believe every Oregonian should have access to the medications they need without undue financial burden,” according to John Mullin, president of the Oregon Coalition for Affordable Prescriptions.
“We firmly believe every Oregonian should have access to
the medications they need without undue financial burden.”
 State Drug Price Review
State Drug Price Review
Oregon is one of nine states looking at drug affordability, with more eyeing the possibility. Senator Deb Patterson, D-Salem, who pushed to create the board in the 2021 legislative session, told The Lund Report, “Oregonians need to have information on what it costs to purchase prescription drugs and they need help to afford … life-saving medications.”
The legislation creating the board initially gave it the power to cap drug prices, but pharmaceutical industry pressure led to that provision being dropped. The 2023 Oregon legislature did pass a board recommendation to cap the out-of-pocket cost of insulin at $35.
Among the claims made by pharmaceutical industry lobbyists is that the board has failed to consider health equity as well as the effect of discounts and rebates on drug affordability. Industry representatives said the board should be more proactive in seeking the views of patients and caregivers.
 The Lund Report identified two local patient groups that have criticized the board’s drug price review process. Lorren Sandt, executive director of Caring Ambassadors Program, Inc. in Oregon City, said price caps on some drugs could lead to price increases for other drugs.
The Lund Report identified two local patient groups that have criticized the board’s drug price review process. Lorren Sandt, executive director of Caring Ambassadors Program, Inc. in Oregon City, said price caps on some drugs could lead to price increases for other drugs.
In an interview, Megan O’Reilly, a national AARP vice president for government affairs, defended state boards as “another tool in our toolbox to lower prescription drug prices at the state level.” Five states have given their boards the ability to set prices for some drugs. Colorado became the first to impose a price cap after its board declared Enbrel, a rheumatoid arthritis drug, unaffordable.
The Colorado board found the average annual cost of Enbrel in 2022 was $46,000, with a patient out-of-pocket cost totaling $2,295. The board’s action touched off a legislative effort to undo the price cap. In a Securities Exchange Commission filing, Amgen, which makes Enbrel, cited Colorado’s price cap as having the potential to “negatively affect” product sales.
Federal Drug Price Negotiations
The Inflation Reduction Act of 2022 gave Medicare the authority to negotiate lower prescription drug prices. The first 10 drugs were announced last August and negotiated prices could go into effect January 1, 2026. Maximum prices were published last month.
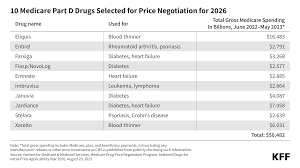
The 10 drugs selected for the first round of negotiations include treatments for several medical conditions, including diabetes (Farxiga, Fiasp/NovoLog, Januvia, Jardiance), blood clots (Eliquis, Xarelto), heart failure (Entresto, Farxiga), psoriasis (Stelara, Enbrel), rheumatoid arthritis (Enbrel), Crohn’s disease (Stelara), and blood cancers (Imbruvica).
Another 15 drugs are scheduled for negotiation and price reductions in 2027, another 15 for price cuts in 2028 and 20 more for 2029 and beyond.
 Drugs qualifying for the first round of Medicare negotiation must be covered under Medicare Part D, Medicare’s outpatient prescription drug benefit program, and are single source brand-name drugs or biological products without therapeutically equivalent generic or biosimilar alternatives approved or licensed. To be eligible for negotiation for 2026, a drug product must have been approved on or before September 1, 2016, and a biological product must have been licensed on or before September 1, 2012.
Drugs qualifying for the first round of Medicare negotiation must be covered under Medicare Part D, Medicare’s outpatient prescription drug benefit program, and are single source brand-name drugs or biological products without therapeutically equivalent generic or biosimilar alternatives approved or licensed. To be eligible for negotiation for 2026, a drug product must have been approved on or before September 1, 2016, and a biological product must have been licensed on or before September 1, 2012.
The initial 10 drugs marked for negotiation were chosen from a list of 50 medications based on how much Medicare spent on them. Data shows Medicare Part D paid more than $50 billion for those drugs between June 2022 and May 2023.
Drug Prices and Pharmacy Benefit Managers
Drug pricing is entangled with the role and efficacy of pharmacy benefit managers, a topic lawmakers took up again in the 2024 session.
Drug makers have pointed a finger at pharmacy benefit managers that negotiate prices on behalf of health insurers with drug manufacturers and pharmacies and process drug claims. Pharmacy benefit managers have been criticized for retaining some or all of drug rebates from manufacturers.
The industry also is under fire because three firms control nearly 80 percent of the pharmacy benefit manager market. One of the largest pharmacy benefit managers just merged with a large health insurer, but many are tightly affiliated with insurers.
Some pharmacy benefit managers operate pharmacies, even as they are blamed for driving many pharmacies, especially in rural areas, out of business.

Board Access to Data
Judge Mosman’s order issued last month mostly sides with PhRMA argument, but leaves intact drug manufacturer reporting requirements for drugs covered by patient assistance programs that experience a 10 percent increase from the previous year.
Drug makers won’t have to report “percent price increase, reasons for the price increase, research and development costs supported by public funds, direct costs related to manufacturing, marketing, distribution, and ongoing research, sales revenue, profit, and prices paid in other countries for the drug.”
Jonathan Bartholomew, AARP director of government affairs in Oregon, said the board will have “plenty of information” to do its work, including claims databases and other sources.




